Good question, I've myself felt very unsure about these things, time to dig a little deeper and perhaps get some basic understanding.
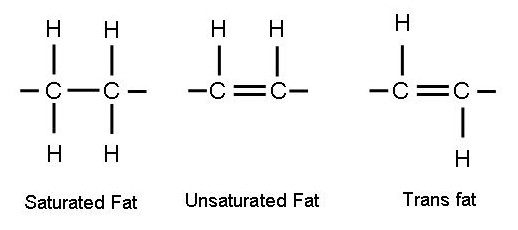
Fats consists of chains of carbon atoms, hydrogen atoms and some oxygen atoms. Two carbon atoms are "joined", or connected to each other by sharing some outer shell electrons.
If the carbon atoms join by sharing two electrons, it's a "single bond".
If the carbon atoms are joined by sharing four electrons it's a "double bond"
A single bond means greater flexibity in how the carbon atoms can move in regards to each other, it's only one link and less stiff than if it's a double bond with two bonds with four electrons. That's why unsaturated fats are liquid at room temperature and saturated fats are not.
Unsaturated fats have one or more double bonds, which means there are more electrons swishing arout out there in the "outer orbit" electron cloud than in saturated fats, which only have single bonds.
All those electrons flying around there in the unsaturated fat makes it highly reactive, these electrons can easily catch up some passing by hydrogen atom. The fat goes rancid.
If you add more hydrogen atoms, thus reducing the number of electrons roaming around, it gets more solid.
Which is what you do when you transform an unsaturated fat to "partially hydrogenerated" by adding a lot of hydrogen to it. You heat it up and press hydrogen into it under pressure. It became popular during WWII with all these logistical problems with supporting large armies and keepin everyone reasonably happy.
Trans fats is solid at room temperature, it gives food manufacturers the ability to create tasty products that be stored at room temperature without getting smelly, with structure enough for packing and and distribution.
The problem with these hydrogenerated, or trans fats, comes from the fact that you don't find much of them in nature. Our bodies have evolved to deal with saturated fat since we love bacon, and unsaturated fat since we sometimes find nuts and avocados.
The hydrogenerated fat is treated like it's saturated fat, but it's not. The process to use it for energy at a cellular level is not very efficient, it's stored somewhere making you fat, which is bad, or in your arteries, which isn't all that great either.
Adding some hydrogen to these unsaturated fats, makes them move in a different way from both saturated and unsaturated fats. Unsaturated fats are very pliable, they can easily move through all of your 100.000 km of blood vessels, dissolving plack along the way. Hydrogenerated fats are not as pliable, and doesn't dissolve much plack, instead they get stuck in smaller blood vessels and create more of it.
I'm not sure how dangerous these trans fats actually are, but I see no reason other than convenience, poverty or laziness to eat them.
No margarine, no cookies in brightly coloured packages from stores, no stock cubes (?), avoid ready meals, don't eat too often at inexpensive restaurants since they stopped cooking a long time ago, they use cheap labour to quickly put together factory made products from these brightly coloured packages nowadays. Make your own food from scratch if possible, put it in a box and bring it to work.
With this diet, and especially with the low carb thing, you get a lot of your energy from fat in general. I would suppose the quality of these fats gets more important.
Some links,
Not sure about her views un saturated fats, but an excellent explanaition if you have 10 minutes,
http://www.youtube.com/watch?v=AgjxPH_jaJk#t=601http://www.headheritage.co.uk/uknow/features/?id=19http://www.dummies.com/how-to/content/saturated-monounsaturated-and-polyunsaturated-fats.htmlhttp://wewantorganicfood.com/2007/09/02/saturated-fats-versus-partially-hydrogenated-vegetable-oils-and-trans-fats/Nice pics of the molecular structure of various fats,
http://www.hhmi.org/biointeractive/molecular-structure-fat, picture 11 and 12 explains an interesting difference, how a trans fat fatty acid is straight instead of slightly bent. I could imagine it getting stuck in some capillary in your brain.
OK, that's about what I can make of it, please correct me if I got something wrong here.